我们决定向甘孜泸定、雅安石棉捐赠300万元现金、300万元物资,目前已成功对接甘孜州红十字会、雅安市红十字会,今天下午已经完成打款,物资根据当地所需正在紧急集结。对于灾区需要的其他支持,我们也当全力以赴。”
9月6日下午,四川科伦药业股份有限公司相关负责人告诉记者,针对四川泸定6.8级地震中受灾严重的泸定县和石棉县,他们紧急启动灾害应急处理方案,并进行现金和物资捐赠。
2026-02-06
CHENGDU, China, February 4, 2026—Sichuan Kelun-Biotech Biopharmaceutical Co., Ltd. ("Kelun-Biotech" or the "Company", 6990.HK) announced that a new indication application for its TROP2-directed ADC sacituzumab tirumotecan (sac-TMT, also known as SKB264/MK-2870) (佳泰莱®) has been approved by the National Medical Products Administration (NMPA) of China for treatment of adult patients with unresectable or metastatic HR+/HER2- (IHC 0, IHC 1+ or IHC 2+/ISH-) breast cancer (BC) who have received prior endocrine therapy (ET) and at least one line of chemotherapy in advanced setting. This approval for HR+/HER2- BC after at least one prior line of chemotherapy marks the fourth indication for sac-TMT approved for marketing in China.
The approval is based on the positive results from the Phase III OptiTROP-Breast02 study which was selected as a Late-Breaking Abstract (LBA) and presented as an oral report at the 2025 European Society for Medical Oncology (ESMO) Congress.
The OptiTROP-Breast02 study evaluated the efficacy and safety of sac-TMT monotherapy compared to investigator's choice of chemotherapy in patients with unresectable or metastatic HR+/HER2- BC. Of the patients enrolled in this Phase III study, 95.7% had visceral metastases, 75.9% had liver metastases; 52.9% were HER2-zero (IHC 0), while 47.1% were HER2-low (IHC 1+ or IHC 2+/ISH-). All patients had received prior CDK4/6 inhibitor and taxane therapy; 56.6% had received ≥2 lines of prior chemotherapy in the advanced or metastatic setting.
Results showed that sac-TMT demonstrated a statistically significant and clinically meaningful increase in progression-free survival (PFS) as assessed by the Blinded Independent Central Review (BICR) compared to chemotherapy (8.3 vs. 4.1 months; hazard ratios (HR), 0.35; 95% CI: 0.26-0.48; p<0.0001). Consistent PFS benefits were observed across all pre-specified subgroups, including HER2-zero and HER2-low, number of chemotherapy lines received in the advanced or metastatic setting, presence of baseline visceral and liver metastases and previous CDK4/6 inhibitor use. According to BICR-assessed PFS results, the hazard ratios in the HER2-zero and HER2-low (IHC 1+ or IHC 2+/ISH-) subgroups were 0.39 (95% CI: 0.26-0.57) and 0.31 (95% CI: 0.20-0.48), respectively. A trend towards overall survival (OS) benefit and a significantly higher objective response rate (ORR) (41.5% vs. 24.1%) were also observed compared with chemotherapy. [1]
Currently, Phase III clinical studies of sac-TMT with or without pembrolizumab (KEYTRUDA®[2]) for the treatment of chemotherapy-naïve HR+/HER2- BC who have received prior ET have been initiated globally (NCT06312176) and in China (NCT07071337).
About HR+/HER2- Breast Cancer
Breast cancer is one of the most common malignant tumors that seriously threaten women's health worldwide. In 2022, there were about 2,297,000 new cases of breast cancer and 666,000 deaths worldwide. Among them, HR+/HER2- breast cancer is the most common subtype, accounting for about 70% of all breast cancer cases, and advanced HR+/HER2- breast cancer has a poor prognosis. This subtype is typically sensitive to hormonal therapy, and therefore, endocrine therapy combined with a CDK4/6 inhibitor constitutes the standard treatment. However, for patients with HR+/HER2- advanced breast cancer whose disease progresses on endocrine therapy, chemotherapy is widely used in clinical while it is associated with low response rate (ORR approximately 14%-22.9%) and limited survival benefit (mPFS approximately 4.0-4.9 months).
About Sac-TMT
Sac-TMT, a core product of the Company, is a novel human TROP2 ADC in which the Company has proprietary intellectual property rights, targeting advanced solid tumors such as non-small cell lung cancer (NSCLC), breast cancer (BC), gastric cancer (GC), gynecological tumors, among others. Sac-TMT is developed with a novel linker to conjugate the payload, a belotecan-derivative topoisomerase I inhibitor with a drug-to-antibody-ratio (DAR) of 7.4. Sac-TMT specifically recognizes TROP2 on the surface of tumor cells by recombinant anti-TROP2 humanized monoclonal antibodies, which is then endocytosed by tumor cells and releases the payload KL610023 intracellularly. KL610023, as a topoisomerase I inhibitor, induces DNA damage to tumor cells, which in turn leads to cell-cycle arrest and apoptosis. In addition, it also releases KL610023 in the tumor microenvironment. Given that KL610023 is membrane permeable, it can enable a bystander effect, or in other words kill adjacent tumor cells.
In May 2022, the Company licensed the exclusive rights to MSD (the tradename of Merck & Co., Inc, Rahway, NJ, USA) to develop, use, manufacture and commercialize sac-TMT in all territories outside of Greater China (which includes Mainland China, Hong Kong, Macao and Taiwan).
To date, four indications for sac-TMT have been approved and marketed in China for: EGFR mutant-positive locally advanced or metastatic non-squamous NSCLC following progression on EGFR-TKI therapy and platinum-based chemotherapy; unresectable locally advanced or metastatic TNBC who have received at least two prior systemic therapies (at least one of them for advanced or metastatic setting); EGFR mutant-positive locally advanced or metastatic non-squamous NSCLC who progressed after treatment with EGFR-TKI therapy; unresectable or metastatic HR+/HER2- (IHC 0, IHC 1+ or IHC 2+/ISH-) BC who have received prior ET and at least one line of chemotherapy in advanced setting. The first two indications listed above have been included in China's National Reimbursement Drug List (NRDL). This inclusion is expected to bring clinical benefits to a greater number of patients with BC and NSCLC. Additionally, sac-TMT has been granted six Breakthrough Therapy Designations (BTDs) by the NMPA.
Sac-TMT is the world's first TROP2 ADC drug approved for marketing in lung cancer. As of today, Kelun-Biotech has initiated 9 registrational clinical studies in China. MSD is evaluating16 ongoing Phase III global clinical studies of sac-TMT as a monotherapy or with pembrolizumab or other anti-cancer agents for several types of cancer. These studies are sponsored and led by MSD.
About Kelun-Biotech
Kelun-Biotech (6990.HK) is a holding subsidiary of Kelun Pharmaceutical, which focuses on the R&D, manufacturing, commercialization and global collaboration of innovative biological drugs and small molecule drugs. Kelun-Biotech focuses on major disease areas such as solid tumors, autoimmune, and metabolic diseases, and in establishing a globalized drug development and industrialization platform to address the unmet medical needs in China and the rest of world. Kelun-Biotech is committed to becoming a leading global enterprise in the field of innovative drugs. At present, Kelun-Biotech has more than 30 ongoing key innovative drug projects, of which 4 projects have been approved for marketing, more than 10 projects are in the clinical stage. Kelun-Biotech has established one of the world's leading proprietary ADC and novel DC platforms, OptiDC™, and has 2 ADC projects approved for marketing, and multiple ADC and novel DC assets in clinical or preclinical research stage. For more information, please visit https://en.kelun-biotech.com/.
Reference Sources:
[1] Fan Y, Li H, Wang H, et al. ESMO Congress 2025, LBA23.
[2] KEYTRUDA® (pembrolizumab) is a registered trademark of Merck Sharp & Dohme LLC (MSD), a subsidiary of Merck & Co., Inc., Rahway, NJ, USA.
2026-01-16
CHENGDU, China, Jan. 16, 2026 /PRNewswire/ -- From January 12 to 15, 2026, the 44th J.P. Morgan Healthcare Conference (JPMHC) was held in San Francisco, California, USA. Dr. Ge Michael, President and CEO of Sichuan Kelun-Biotech Biopharmaceutical Co., Ltd. ("Kelun-Biotech" or the "Company", 6990.HK), was invited to attend the conference and delivered a keynote speech on the morning of January 15 (local time) and presented the Company's latest achievements in drug R&D, commercialization, and globalization, and outlined its innovation strategy and future development plans.
Since initiating its innovation journey in 2012, Kelun-Biotech has rapidly emerged as a leader in China's innovative drug field, building differentiated technology platforms and robust R&D pipelines. Leveraging its global leading OptiDC™ platform for antibody-drug conjugates (ADCs) and novel drug conjugates (DCs), the Company continuously advances the differentiated development of ADCs and novel DCs, forming a gradient portfolio for treating multiple tumor types. Currently, Kelun-Biotech has two ADC products on market: sacituzumab tirumotecan (sac-TMT, 佳泰莱®) and trastuzumab botidotin (舒泰莱®), covering breast cancer and lung cancer indications. Additionally, nine uniquely designed ADC and novel DC drugs—including cutting-edge directions such as bispecific ADC and radiopharmaceutical conjugate (RDC) are in clinical stage. For high-incidence tumor types in China, such as breast cancer, lung cancer, and gastrointestinal tumors, the Company has initiated nine pivotal studies, while multiple Phase II clinical studies targeting gynecological tumors are progressing steadily. Furthermore, Kelun-Biotech has also developed several non-DC candidates and expanded indications into non-oncology areas.
Over the past year, Kelun-Biotech has presented multiple research findings at international academic conferences and published in authoritative journals: three were selected for oral presentations at the American Society of Clinical Oncology (ASCO) annual meeting, and three were selected as Late-Breaking Abstracts (LBA) for oral presentations at the European Society for Medical Oncology (ESMO) Congress. Among these, the results of the OptiTROP-Lung04 study of sac-TMT for treating EGFR-mutant Non-Small Cell Lung Cancer (NSCLC) after TKI therapy were presented at the Presidential Symposium session of ESMO and simultaneously published in The New England Journal of Medicine, highlighting its global academic and clinical value.
In terms of commercialization, Kelun-Biotech has formed a competitive initial product portfolio. Its core product, the TROP2-directed ADC sac-TMT, has been approved in China for three indications: second-line and above triple-negative breast cancer, second-line and third-line EGFR-mutated NSCLC. The HER2-directed ADC trastuzumab botidotin was approved last year for second-line and above HER2-positive breast cancer, becoming the first domestically developed HER2-directed ADC approved for this indication. Furthermore, the anti-EGFR monoclonal antibody Cetuximab N01 (达泰莱®) for RAS wild-type colorectal cancer and the anti-PD-L1 monoclonal antibody tagitanlimab (科泰莱®) for nasopharyngeal carcinoma have been launched. Another small-molecule RET inhibitor A400 is expected to be approved within the year, bringing the total number of commercialized products in China to five. Currently, three of the Company's commercialized products, covering five indications, have been included in the National Reimbursement Drug List (NRDL), further benefiting a broad population of cancer patients.
While strengthening its presence in the domestic market, Kelun-Biotech is actively expanding overseas. It has established collaborations with MSD, Ellipses, Windward Bio and Crescent Biopharma to maximize the value of its pipeline value and corporate worth. Among these, MSD is evaluating 16 global Phase III clinical studies of sac-TMT.
The breakthroughs in both clinical development and commercialization are driven by the Company's sustained investment in innovative R&D. With over a decade of accumulation in the ADC field, Kelun-Biotech's proprietary OptiDC™ platform enables differentiated design of drug candidates, which combines specific targets or targeting mechanisms with the most suitable payload-linker strategy to balance efficacy and safety. Additionally, the company is adopting a "multi-pronged" innovation strategy to continuously enhance platform capabilities. By exploring novel targets, new payloads, and diverse conjugation technologies, it is expanding the application boundaries across both oncology and non-oncology fields.
Looking ahead, Kelun-Biotech will consolidate its foundations in R&D, technologies, platforms, and operations by Executing five key development strategies. Concurrently, the Company will elevate its globalization strategy, enhancing its capabilities in product development, registration, and commercialization in ex-China market, advancing on its path to becoming a world-class biopharmaceutical company.
Please refer to the "Investor Relations-Investor Calendar" page of the company's official website for the presentation materials. You can visit the website for further details.
About Kelun-Biotech
Kelun-Biotech (6990.HK) is a holding subsidiary of Kelun Pharmaceutical (002422.SZ), which focuses on the R&D, manufacturing, commercialization and global collaboration of innovative biological drugs and small molecule drugs. Kelun-Biotech focuses on major disease areas such as solid tumors, autoimmune, and metabolic diseases, and in establishing a globalized drug development and industrialization platform to address the unmet medical needs in China and the rest of world. Kelun-Biotech is committed to becoming a leading global enterprise in the field of innovative drugs. At present, Kelun-Biotech has more than 30 ongoing key innovative drug projects, of which 4 projects have been approved for marketing, 1 project is in the NDA stage and more than 10 projects are in the clinical stage. Kelun-Biotech has established one of the world's leading proprietary ADC and novel DC platforms, OptiDC™, and has 2 ADC projects approved for marketing, and multiple ADC and novel DC assets in clinical or preclinical research stage. For more information, please visit https://en.kelun-biotech.com/.
2024-03-19
More

(“Kelun-Biotech” or the “Company”, SEHK: 6990.HK), an innovative TROP2-ADC (SKB264, MK-2870), a randomized, open, multicenter study to evaluate the efficacy of SKB264 in the first-line treatment of chemotherapy in patients with recurrent or metastatic non-resectable triple-negative breast cancer (TNBC) in advanced stages. SKB264 comparing investigator-selected chemotherapy for the first-line treatment of patients with non-surgically resectable recurrent or metastatic triple-negative breast cancer (TNBC) to a randomized, open, multicenter Phase III registry-based clinical study in patients with advanced-stage, non-systemic-treatment-na?ve PD-L1-negative TNBC (the “OptiTROP Breast03 (“OptiTROP Breast03” or “the Study”) was successfully held in Hefei.

This study a clinical trial evaluating SKB264 comparing investigator-selected chemotherapy first-line for the treatment of patients with non-operable resectable recurrent or metastatic triple-negative breast cancer (Protocol No. SKB264-III-11), with the national lead research center being the Affiliated Cancer Hospital of Fudan University, the lead researcher being Prof. Zhi-Min Shao, and the clinical registry numbers being: CTR20240413, NCT06279364, and is currently being rapidly pushed into the group.

Professor Zhimin Shao, Chairman of the conference, said, “It is a great honor to lead the registrational clinical study of ColumboTech SKB264 in triple negative breast cancer. Currently, TNBC is a hot area of clinical research. We believe that based on the excellent performance of TROP2 ADC SKB264 reported in first-line as well as second-line and above treatment of advanced TNBC in triple-negative breast cancer in the previous phase, SKB264 can achieve better clinical results in the first-line treatment of triple-negative breast cancer in China. We sincerely thank all the experts for their active participation in this trial, and hope that through our joint efforts and professional wisdom, we can make positive contributions to this clinical study, and look forward to SKB264 receiving marketing approval as soon as possible for the benefit of more patients”.

Dr. Ge Junyou shared, “Kelun is the world's largest manufacturer of infusion solutions in terms of production scale, the world's largest manufacturer of antibiotic intermediates in terms of production scale, and a leading head enterprise of pharmaceutical innovation in China. Kelun-Biotech carries the champion gene of the Group and adheres to innovation at source.SKB264 is the first TROP2 ADC-like drug in China to enter Phase III and enter NDA registration.SKB264's NDA filing for TNBC backline treatment has been accepted by the State Drug Administration, and the drug has been recognized as a breakthrough therapy by four organizations, and has initiated a number of clinical studies in different indications in addition to TNBC. The drug has also received four breakthrough therapeutic recognitions. We look forward to working together to rapidly advance the OptiTROP Breast03 study and accelerate the marketing process for this indication, which will benefit more breast cancer patients. Thank you for the great support from all the experts!”

Professor Cao Min from Hunan Cancer Hospital shared his detailed experience in terms of center overview and enrollment, case sharing, screening and enrollment process management, and internal and external multi-party collaboration.

Professor Liang Yan from Jiangsu Provincial People's Hospital shared detailed experience from center enrollment, case sharing, subjects in group management and management of common adverse events.

Professor Fan Lei from Fudan University Cancer Hospital gave a detailed presentation on the program design and implementation details of the project.

Under the chairmanship of Prof. Zhimin Shao and Dr. Xiaoping Jin, Chief Medical Officer of Clonbuterol, all the participating experts had a full and enthusiastic discussion on the study design, enrollment criteria of the study population, and the study process, and reached a consensus.

Professor Wang Zhonghua and Prof. Shao Zhimin from the Affiliated Cancer Hospital of Fudan University summarized the meeting at the end and thanked all the experts who participated in the investigator meeting. The investigators' meeting came to a successful conclusion, officially starting a brand-new journey for the OptiTROP Breast03 study. Clonbuterol has been exploring the advantageous areas of ADC technology in depth, and continues to promote the deepening layout of SKB264 clinical research. SKB264 first-line treatment of non-surgically resectable locally advanced, recurrent or metastatic PD-L1 TNBC has also been recognized by the BTD, and Clonbuterol will efficiently and qualitatively complete the registry study, and make joint efforts with the first-line clinical force to bring more hope and gospel to global tumor patients. Gospel.
About Kelun-Biotech:
Kelun-Biotech(6990.HK)is a holding subsidiary of Kelun Pharmaceutical (002422.SZ), which focuses on the R&D, manufacturing, commercialization and global collaboration of innovative biological drugs and small molecule drugs. The company focuses on major disease areas such as solid tumors, autoimmune, inflammatory, and metabolic diseases, and in establishing a globalized drug development and industrialization platform to address the unmet medical needs in China and the rest of world. The Company is committed to becoming a leading global enterprise in the field of innovative drugs.
At present, the Company has more than 30 ongoing innovative projects in major disease areas such as solid tumors, autoimmune, inflammatory, and metabolic diseases, including over 10 projects in the clinical stage and 4 projects in the NDA stage with several global trials being conducted simultaneously in multiple countries, including China, Europe, and the United States. The company has established one of the world’s leading proprietary ADC platforms, OptiDC?, and has 5 ADC projects in the clinical stage (2 of which are in the NDA stage) and several projects in the preclinical stage. For more information, please visit https://kelun-biotech.com/.
2024-03-14
More
Micro-world at your fingertips, customized nature :
“In this season of spring blossoming and grass growing, during the “Goddess Month” in March, the company issued holiday gifts and presents to all female employees to send holiday greetings and best wishes, and organized a March March Goddess Month handmade activities.

One bottle, one world, Kelun-Biotech’s micro landscape eco-bottle DIY activity took female employees to put the warmth of greenery into glass bottles, feel the power of plants together, and add a touch of fresh fun to life.
Crystal clear glass bottles, green moss plants, staggered landscape fabrics, fantasy cartoon dolls... In the fun atmosphere, everyone made the bright spring mood into unique limited edition works.

This handcraft activity not only stimulated everyone's curiosity about things in nature and knowledge of ferns and mosses, but also allowed everyone's creative thinking to be displayed in an exclusive space, conveying the concept of living with love for greenery and the environment.
Ancient style lantern DIY, enjoy the thousand years of cultural customs:
Lantern is one of the typical elements of China's traditional culture and a symbol of good luck. This month, Kelun-Biotech carried out the DIY activity of ancient style lanterns, leading the staff to appreciate the millennium culture and customs.

This activity is a traditional and modern fusion of cultural feast. Staff do-it-yourself, exquisite ancient style lanterns gradually presented in front of everyone, in the staff to exercise the ability to do it at the same time, but also harvested their own holiday exclusive gift, heritage and promote the excellent traditional culture of the Chinese nation, feel the beauty of the holiday and warmth, effectively enrich the cultural life of the staff, set up a platform for interaction, communication, mutual assistance.

About Kelun-Biotech: Kelun-Biotech(6990.HK)is a holding subsidiary of Kelun Pharmaceutical (002422.SZ), which focuses on the R&D, manufacturing, commercialization and global collaboration of innovative biological drugs and small molecule drugs. The company focuses on major disease areas such as solid tumors, autoimmune, inflammatory, and metabolic diseases, and in establishing a globalized drug development and industrialization platform to address the unmet medical needs in China and the rest of world. The Company is committed to becoming a leading global enterprise in the field of innovative drugs.
At present, the Company has more than 30 ongoing innovative projects in major disease areas such as solid tumors, autoimmune, inflammatory, and metabolic diseases, including over 10 projects in the clinical stage and 4 projects in the NDA stage with several global trials being conducted simultaneously in multiple countries, including China, Europe, and the United States. The company has established one of the world’s leading proprietary ADC platforms, OptiDC?, and has 5 ADC projects in the clinical stage (2 of which are in the NDA stage) and several projects in the preclinical stage. For more information, please visit https://kelun-biotech.com/.
2024-03-13
More
On 8th March 2024, Sichuan Kelun-Biotech Biopharmaceutical Co., Ltd. (6990.HK, Kelun-Biotech)’s innovative core product TROP2-ADC SKB264 (also known as MK-2870) was granted Breakthrough Therapy Designation(BTD) by the Center for Drug Evaluation( CDE) of the National Medical Products Administration (NMPA) of China for first-line treatment of unresectable locally advanced, recurrent or metastatic PD-L1 negative triple negative breast cancer (TNBC). SKB264 (MK-2870) is jointly developed by Kelun-Biotech and MSD (the tradename of Merck & Co., Inc., Rahway, NJ, USA).
This is the fourth Breakthrough Therapy Designation for SKB264 (MK-2870) granted by the NMPA. Previously, SKB264 (MK-2870) was granted BTD for:
July 2022, locally advanced or metastatic TNBC.
January 2023, EGFR-mutant locally advanced or metastatic non-small cell lung cancer (NSCLC) after progression on EGFR tyrosine kinase inhibitor (TKI) therapy.
June 2023, locally advanced or metastatic hormone receptor-positive (HR+) and human epidermal growth factor receptor 2-negative (HER2-) breast cancer in patients who have previously received at least two lines of systematic chemotherapy.
Breast cancer is one of the most common malignant tumors in women, with its incidence showing an increasing trend year by year. The International Agency for Research on Cancer (IARC) and the World Health Organization (WHO) estimated that in 2020, there were over 2.26 million new cases of breast cancer globally, accounting for 11.7% of all tumors, making it the most prevalent cancer [1]. Triple-negative breast cancer (TNBC) is a clinical subtype that is negative for estrogen receptor (ER), progesterone receptor (PR), and human epidermal growth factor receptor 2 (HER2), comprising 10.0% to 20.8% of all breast cancer types. Triple-negative breast cancer is characterized by high heterogeneity, a high rate of recurrence and metastasis, and a poorer prognosis compared to other types of breast cancer. It lacks effective and specific treatment methods [2]. Currently, the primary treatment for PD-L1 negative TNBC is chemotherapy, which offers limited survival benefits, highlighting the urgent need for new treatment plans. For patients with inoperable locally advanced, recurrent, or metastatic PD-L1 negative triple-negative breast cancer, the first-line standard treatment in China still involves chemotherapy, including single-agent or combination chemotherapy [3]. Compared to single-agent chemotherapy, combination chemotherapy has greater toxicity and offers limited survival benefits.
BTD is designed to expedite the development of new drugs for serious diseases that have shown significant efficacy or safety over existing therapies in preliminary clinical trials. For new drugs included in the Breakthrough Therapy List, CDE will prioritize the allocation of resources for communication, enhanced guidance, and promotion of drug development. If the relevant conditions have been assessed to be met, an application for conditional approval and an application for priority review and approval may also be submitted at the time of the application for drug marketing authorization. This will help accelerate the SKB264 (MK-2870) development process and address the unmet clinical needs of Chinese patients as soon as possible.
参考文献:
[1] Cancer statistics in China and United States, 2022: profiles, trends, and determinants. Chin Med J(Engl).2022 Feb 9; 135(5):584-590.
[2] 三阴性乳腺癌含铂方案临床应用专家共识(2021版).中华肿瘤防治杂志2021年6月第28卷第12期.
[3] 《中国临床肿瘤学会(CSCO)乳腺癌诊疗指南2023版》.
2024-03-13
More
Our key product A400(EP0031), a small molecule rearranged during transfection (RET) kinase inhibitor program, has been granted Fast Track designation by the United States Food and Drug Administration (FDA) for the treatment of RET fusion-positive non-small cell lung cancer (NSCLC).

A400 (EP0031) is a second-generation selective RET inhibitor (SRI) with broad activity against common RET fusions and mutations.
In March 2021, the Company granted Ellipses Pharma Limited, a U.K.-based international drug development company, an exclusive, royalty-bearing, sub licensable license to develop, manufacture and commercialize A400 (EP0031) in all countries excluding Greater China, North Korea, South Korea, Singapore, Malaysia and Thailand.
In June 2022, the FDA approved an investigational new drug application for A400 (EP0031), and a phase 1/2 trial is ongoing in patients with malignant tumors with RET gene alteration.
In November 2023, A400 was granted Orphan Drug Designation by the FDA for the treatment of RET fusion-positive solid tumors.
In preclinical studies, A400 (EP0031) demonstrated favourable inhibitory activity against key RET kinases in-vitro and in-vivo. A400 (EP0031) also demonstrated good penetration of the blood brain barrier in animal models. Data shared at the 2023 American Society of Clinical Oncology (ASCO) Annual Meeting on A400 (EP0031) showed promising anti-tumor efficacy in patients with advanced RET+ solid tumors, highlighted by ORR of 80.8% and 69.7% for 1L and 2L+ advanced RET+ NSCLC, respectively, based on results from its ongoing phase 1/2 trial. In both cases, DCR of over 96% were reported.
At present, the Company is conducting A400 (EP0031) pivotal clinical study in China for RET- positive NSCLC.
About Ellipses Pharma Limited
Ellipses Pharma is a global drug development company based in London, focused on accelerating the development of cancer treatments through an innovative drug development model that combines unbiased vetting to de-risk initial asset selection with an uninterrupted funding flow to minimise the time it takes to advance lead products through clinical trials and reach patients.
About Kelun-Biotech
Kelun-Biotech(6990.HK)is a holding subsidiary of Kelun Pharmaceutical (002422.SZ), which focuses on the R&D, manufacturing, commercialization and global collaboration of innovative biological drugs and small molecule drugs. The company focuses on major disease areas such as solid tumors, autoimmune, inflammatory, and metabolic diseases, and in establishing a globalized drug development and industrialization platform to address the unmet medical needs in China and the rest of world. The Company is committed to becoming a leading global enterprise in the field of innovative drugs. At present, the Company has 33 ongoing innovative projects in major disease areas such as solid tumors, autoimmune, inflammatory, and metabolic diseases, including 14 projects in the clinical stage with several global trials being conducted simultaneously in multiple countries, including China, Europe, and the United States. The company has established one of the world’s leading proprietary ADC platforms, OptiDC, and has four ADC projects in the clinical stage (two of which are in the NDA stage) and several projects in the preclinical stage. For more information, please visit https://kelun-biotech.com/.
2024-03-04
More
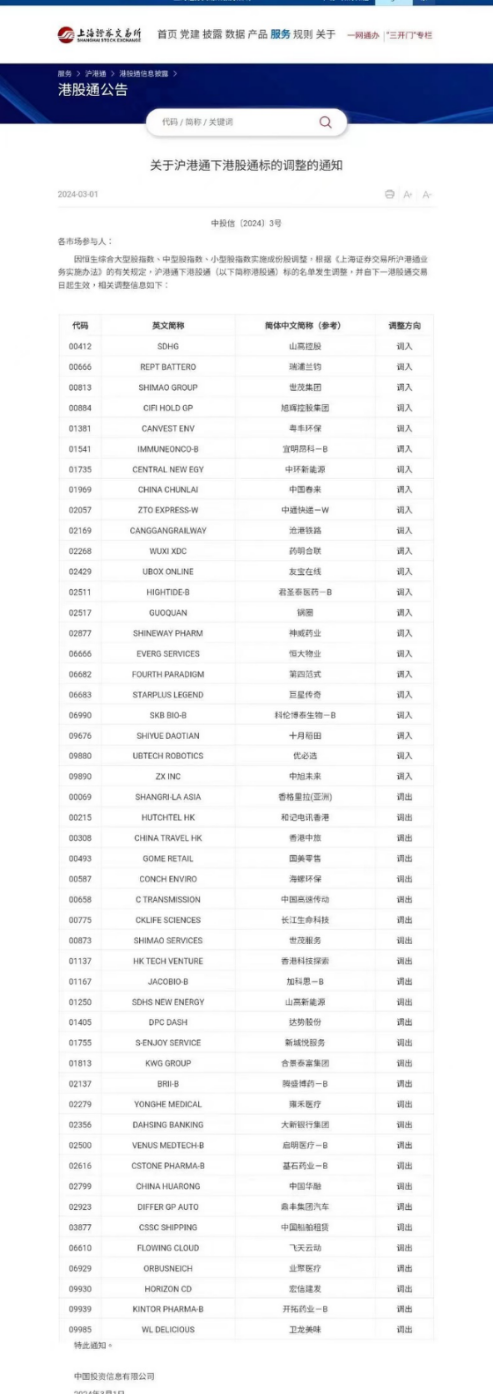
pursuant to the Notice of Adjustment of Stock List of Southbound Trading Link of the Shanghai-Hong Kong Stock Connect ( 关于沪港通下港股通目标调整的通知) made by the Shanghai StockExchange on March 1, 2024, the Company will be included in the list of eligible shares of the Southbound Trading Link of the Shanghai-Hong Kong Stock Connect with effect from March 4, 2024.
2024-02-26
More

On February 21, 2024, the prestigious international finance magazine, FinanceAsia, held its 2023 annual achievement awards ceremony in Hong Kong. Kelun-Biotech (6990.HK) was invited to attend the celebration dinner and was awarded the "Best IPO in Asia and Hong Kong for 2023" honor.

FinanceAsia is regarded as one of the most representative professional monthly magazine in Asia's capital market and has significant influence in the investment industry. Founded in 1996, its sponsored "FinanceAsia Achievement Awards" is one of the most influential and credible industry award selections in the Asia-Pacific region, the award ceremony is an significant annual gathering in the investment industry.
About Kelun-Biotech
Kelun-Biotech(6990.HK)is a holding subsidiary of Kelun Pharmaceutical (002422.SZ), which focuses on the R&D, manufacturing, commercialization and global collaboration of innovative biological drugs and small molecule drugs. The company focuses on major disease areas such as solid tumors, autoimmune, inflammatory, and metabolic diseases, and in establishing a globalized drug development and industrialization platform to address the unmet medical needs in China and the rest of world. The Company is committed to becoming a leading global enterprise in the field of innovative drugs. At present, the Company has 33 ongoing innovative projects in major disease areas such as solid tumors, autoimmune, inflammatory, and metabolic diseases, including 14 projects in the clinical stage with several global trials being conducted simultaneously in multiple countries, including China, Europe, and the United States. The company has established one of the world’s leading proprietary ADC platforms, OptiDC, and has four ADC projects in the clinical stage (two of which are in the NDA stage) and several projects in the preclinical stage. For more information, please visit https://kelun-biotech.com/.
2024-02-24
More
Clinical trials in non-small cell lung cancer (NSCLC) and endometrial cancer (EC) conducted by Kelun-Biotech and Merck Sharp & Dohme for the core product of the collaboration, SKB264 (MK-2870), have triggered relevant clinical milestone payments totaling $75.0 million. The Company has received a portion of the payment from Merck Sharp & Dohme and expects to receive the balance in the near future.
Collaboration Update:
In May 2022, Kelun-Biotech granted Merck Sharp & Dohme exclusive rights to develop, manufacture and commercialize SKB264 (MK-2870) in all territories outside of Greater China.
Merck Sharp & Dohme announced that it has initiated three pivotal Phase 3 clinical trials evaluating SKB264 (MK-2870) as a monotherapy for the treatment of previously treated advanced or metastatic non-small cell lung cancer (NSCLC) with epidermal growth factor receptor (EGFR) mutations or other genomic alterations, as a monotherapy for the treatment of endometrial cancer (EC) previously treated with platinum-containing chemotherapy and immunotherapy, and with pabopoetin. ), and in combination with pembrolizumab for metastatic NSCLC with programmed death ligand 1 (PD-L1) expression greater than or equal to 50%.
Based on written confirmation from Merck Sharp & Dohme, clinical trials in NSCLC and EC have triggered related clinical milestone payments totaling $75.0 million. The Company has received a portion of the payment from Merck Sharp & Dohme and expects to receive the balance in the near future.
About SKB264 (MK-2870, trade name: Jatelle)
SKB264 is a representative and innovative ADC targeting TROP2 developed by OptiDC, the internationally renowned ADC development platform of Kelun-Biotech. SKB264 consists of a humanized anti-TROP2 monoclonal antibody with high affinity and targeting properties, which is conjugated to the toxin small molecule T030 (topoisomerase I inhibitor) through the stability-optimized CL2A linker. T030 (topoisomerase I inhibitor) with an average drug-antibody ratio (DAR) of 7.4. The stability of SKB264 is enhanced in two ways, allowing more ADCs to reach tumor cells. On the one hand, the antibody end of the linker uses a methylsulfonylpyrimidine linker that irreversibly couples to the antibody, and on the other hand, T030 contains a methylsulfone structure that binds stably to the toxin end of the linker.SKB264's linker combines both extracellular pH-sensitive cleavage of the tumor cell and intracellular enzymatic cleavage of the tumor cell, which allows for the highly efficient release of the toxin's small molecules and the exertion of an antitumor effect.The activity of T030 is comparable to that of DXd and has a bystander effect. In summary, SKB264 has a triple antitumor effect: first, the pH-sensitive linker can be cleaved to release T030 in the acidic tumor microenvironment; second, SKB264 can be endocytosed into the tumor cell and then cleaved to release T030 by zymotics; finally, T030 can transcend the cell membrane to exert the bystander effect to kill the surrounding tumor cells. Finally, T030 can pass through the cell membrane and exert a “bystander effect” to kill the surrounding tumor cells. Good stability, high affinity antibody, high DAR value, high active toxin, triple anti-tumor effects become the cornerstone of SKB264's potent anti-tumor effect, which has also been initially confirmed in clinical studies.
About Kelun-Biotech:
Kelun-Biotech(6990.HK)is a holding subsidiary of Kelun Pharmaceutical (002422.SZ), which focuses on the R&D, manufacturing, commercialization and global collaboration of innovative biological drugs and small molecule drugs. The company focuses on major disease areas such as solid tumors, autoimmune, inflammatory, and metabolic diseases, and in establishing a globalized drug development and industrialization platform to address the unmet medical needs in China and the rest of world. The Company is committed to becoming a leading global enterprise in the field of innovative drugs. At present, the Company has more than 30 ongoing innovative projects in major disease areas such as solid tumors, autoimmune, inflammatory, and metabolic diseases, including over 10 projects in the clinical stage and 4 projects in the NDA stage with several global trials being conducted simultaneously in multiple countries, including China, Europe, and the United States. The company has established one of the world’s leading proprietary ADC platforms, OptiDC?, and has 5 ADC projects in the clinical stage (2 of which are in the NDA stage) and several projects in the preclinical stage. For more information, please visit https://kelun-biotech.com/.
2024-02-09
More

2022年09月06日
我们决定向甘孜泸定、雅安石棉捐赠300万元现金、300万元物资,目前已成功对接甘孜州红十字会、雅安市红十字会,今天下午已经完成打款,物资根据当地所需正在紧急集结。对于灾区需要的其他支持,我们也当全力以赴。”
9月6日下午,四川科伦药业股份有限公司相关负责人告诉记者,针对四川泸定6.8级地震中受灾严重的泸定县和石棉县,他们紧急启动灾害应急处理方案,并进行现金和物资捐赠。