我们决定向甘孜泸定、雅安石棉捐赠300万元现金、300万元物资,目前已成功对接甘孜州红十字会、雅安市红十字会,今天下午已经完成打款,物资根据当地所需正在紧急集结。对于灾区需要的其他支持,我们也当全力以赴。”
9月6日下午,四川科伦药业股份有限公司相关负责人告诉记者,针对四川泸定6.8级地震中受灾严重的泸定县和石棉县,他们紧急启动灾害应急处理方案,并进行现金和物资捐赠。
2026-02-06
CHENGDU, China, February 4, 2026—Sichuan Kelun-Biotech Biopharmaceutical Co., Ltd. ("Kelun-Biotech" or the "Company", 6990.HK) announced that a new indication application for its TROP2-directed ADC sacituzumab tirumotecan (sac-TMT, also known as SKB264/MK-2870) (佳泰莱®) has been approved by the National Medical Products Administration (NMPA) of China for treatment of adult patients with unresectable or metastatic HR+/HER2- (IHC 0, IHC 1+ or IHC 2+/ISH-) breast cancer (BC) who have received prior endocrine therapy (ET) and at least one line of chemotherapy in advanced setting. This approval for HR+/HER2- BC after at least one prior line of chemotherapy marks the fourth indication for sac-TMT approved for marketing in China.
The approval is based on the positive results from the Phase III OptiTROP-Breast02 study which was selected as a Late-Breaking Abstract (LBA) and presented as an oral report at the 2025 European Society for Medical Oncology (ESMO) Congress.
The OptiTROP-Breast02 study evaluated the efficacy and safety of sac-TMT monotherapy compared to investigator's choice of chemotherapy in patients with unresectable or metastatic HR+/HER2- BC. Of the patients enrolled in this Phase III study, 95.7% had visceral metastases, 75.9% had liver metastases; 52.9% were HER2-zero (IHC 0), while 47.1% were HER2-low (IHC 1+ or IHC 2+/ISH-). All patients had received prior CDK4/6 inhibitor and taxane therapy; 56.6% had received ≥2 lines of prior chemotherapy in the advanced or metastatic setting.
Results showed that sac-TMT demonstrated a statistically significant and clinically meaningful increase in progression-free survival (PFS) as assessed by the Blinded Independent Central Review (BICR) compared to chemotherapy (8.3 vs. 4.1 months; hazard ratios (HR), 0.35; 95% CI: 0.26-0.48; p<0.0001). Consistent PFS benefits were observed across all pre-specified subgroups, including HER2-zero and HER2-low, number of chemotherapy lines received in the advanced or metastatic setting, presence of baseline visceral and liver metastases and previous CDK4/6 inhibitor use. According to BICR-assessed PFS results, the hazard ratios in the HER2-zero and HER2-low (IHC 1+ or IHC 2+/ISH-) subgroups were 0.39 (95% CI: 0.26-0.57) and 0.31 (95% CI: 0.20-0.48), respectively. A trend towards overall survival (OS) benefit and a significantly higher objective response rate (ORR) (41.5% vs. 24.1%) were also observed compared with chemotherapy. [1]
Currently, Phase III clinical studies of sac-TMT with or without pembrolizumab (KEYTRUDA®[2]) for the treatment of chemotherapy-naïve HR+/HER2- BC who have received prior ET have been initiated globally (NCT06312176) and in China (NCT07071337).
About HR+/HER2- Breast Cancer
Breast cancer is one of the most common malignant tumors that seriously threaten women's health worldwide. In 2022, there were about 2,297,000 new cases of breast cancer and 666,000 deaths worldwide. Among them, HR+/HER2- breast cancer is the most common subtype, accounting for about 70% of all breast cancer cases, and advanced HR+/HER2- breast cancer has a poor prognosis. This subtype is typically sensitive to hormonal therapy, and therefore, endocrine therapy combined with a CDK4/6 inhibitor constitutes the standard treatment. However, for patients with HR+/HER2- advanced breast cancer whose disease progresses on endocrine therapy, chemotherapy is widely used in clinical while it is associated with low response rate (ORR approximately 14%-22.9%) and limited survival benefit (mPFS approximately 4.0-4.9 months).
About Sac-TMT
Sac-TMT, a core product of the Company, is a novel human TROP2 ADC in which the Company has proprietary intellectual property rights, targeting advanced solid tumors such as non-small cell lung cancer (NSCLC), breast cancer (BC), gastric cancer (GC), gynecological tumors, among others. Sac-TMT is developed with a novel linker to conjugate the payload, a belotecan-derivative topoisomerase I inhibitor with a drug-to-antibody-ratio (DAR) of 7.4. Sac-TMT specifically recognizes TROP2 on the surface of tumor cells by recombinant anti-TROP2 humanized monoclonal antibodies, which is then endocytosed by tumor cells and releases the payload KL610023 intracellularly. KL610023, as a topoisomerase I inhibitor, induces DNA damage to tumor cells, which in turn leads to cell-cycle arrest and apoptosis. In addition, it also releases KL610023 in the tumor microenvironment. Given that KL610023 is membrane permeable, it can enable a bystander effect, or in other words kill adjacent tumor cells.
In May 2022, the Company licensed the exclusive rights to MSD (the tradename of Merck & Co., Inc, Rahway, NJ, USA) to develop, use, manufacture and commercialize sac-TMT in all territories outside of Greater China (which includes Mainland China, Hong Kong, Macao and Taiwan).
To date, four indications for sac-TMT have been approved and marketed in China for: EGFR mutant-positive locally advanced or metastatic non-squamous NSCLC following progression on EGFR-TKI therapy and platinum-based chemotherapy; unresectable locally advanced or metastatic TNBC who have received at least two prior systemic therapies (at least one of them for advanced or metastatic setting); EGFR mutant-positive locally advanced or metastatic non-squamous NSCLC who progressed after treatment with EGFR-TKI therapy; unresectable or metastatic HR+/HER2- (IHC 0, IHC 1+ or IHC 2+/ISH-) BC who have received prior ET and at least one line of chemotherapy in advanced setting. The first two indications listed above have been included in China's National Reimbursement Drug List (NRDL). This inclusion is expected to bring clinical benefits to a greater number of patients with BC and NSCLC. Additionally, sac-TMT has been granted six Breakthrough Therapy Designations (BTDs) by the NMPA.
Sac-TMT is the world's first TROP2 ADC drug approved for marketing in lung cancer. As of today, Kelun-Biotech has initiated 9 registrational clinical studies in China. MSD is evaluating16 ongoing Phase III global clinical studies of sac-TMT as a monotherapy or with pembrolizumab or other anti-cancer agents for several types of cancer. These studies are sponsored and led by MSD.
About Kelun-Biotech
Kelun-Biotech (6990.HK) is a holding subsidiary of Kelun Pharmaceutical, which focuses on the R&D, manufacturing, commercialization and global collaboration of innovative biological drugs and small molecule drugs. Kelun-Biotech focuses on major disease areas such as solid tumors, autoimmune, and metabolic diseases, and in establishing a globalized drug development and industrialization platform to address the unmet medical needs in China and the rest of world. Kelun-Biotech is committed to becoming a leading global enterprise in the field of innovative drugs. At present, Kelun-Biotech has more than 30 ongoing key innovative drug projects, of which 4 projects have been approved for marketing, more than 10 projects are in the clinical stage. Kelun-Biotech has established one of the world's leading proprietary ADC and novel DC platforms, OptiDC™, and has 2 ADC projects approved for marketing, and multiple ADC and novel DC assets in clinical or preclinical research stage. For more information, please visit https://en.kelun-biotech.com/.
Reference Sources:
[1] Fan Y, Li H, Wang H, et al. ESMO Congress 2025, LBA23.
[2] KEYTRUDA® (pembrolizumab) is a registered trademark of Merck Sharp & Dohme LLC (MSD), a subsidiary of Merck & Co., Inc., Rahway, NJ, USA.
2026-01-16
CHENGDU, China, Jan. 16, 2026 /PRNewswire/ -- From January 12 to 15, 2026, the 44th J.P. Morgan Healthcare Conference (JPMHC) was held in San Francisco, California, USA. Dr. Ge Michael, President and CEO of Sichuan Kelun-Biotech Biopharmaceutical Co., Ltd. ("Kelun-Biotech" or the "Company", 6990.HK), was invited to attend the conference and delivered a keynote speech on the morning of January 15 (local time) and presented the Company's latest achievements in drug R&D, commercialization, and globalization, and outlined its innovation strategy and future development plans.
Since initiating its innovation journey in 2012, Kelun-Biotech has rapidly emerged as a leader in China's innovative drug field, building differentiated technology platforms and robust R&D pipelines. Leveraging its global leading OptiDC™ platform for antibody-drug conjugates (ADCs) and novel drug conjugates (DCs), the Company continuously advances the differentiated development of ADCs and novel DCs, forming a gradient portfolio for treating multiple tumor types. Currently, Kelun-Biotech has two ADC products on market: sacituzumab tirumotecan (sac-TMT, 佳泰莱®) and trastuzumab botidotin (舒泰莱®), covering breast cancer and lung cancer indications. Additionally, nine uniquely designed ADC and novel DC drugs—including cutting-edge directions such as bispecific ADC and radiopharmaceutical conjugate (RDC) are in clinical stage. For high-incidence tumor types in China, such as breast cancer, lung cancer, and gastrointestinal tumors, the Company has initiated nine pivotal studies, while multiple Phase II clinical studies targeting gynecological tumors are progressing steadily. Furthermore, Kelun-Biotech has also developed several non-DC candidates and expanded indications into non-oncology areas.
Over the past year, Kelun-Biotech has presented multiple research findings at international academic conferences and published in authoritative journals: three were selected for oral presentations at the American Society of Clinical Oncology (ASCO) annual meeting, and three were selected as Late-Breaking Abstracts (LBA) for oral presentations at the European Society for Medical Oncology (ESMO) Congress. Among these, the results of the OptiTROP-Lung04 study of sac-TMT for treating EGFR-mutant Non-Small Cell Lung Cancer (NSCLC) after TKI therapy were presented at the Presidential Symposium session of ESMO and simultaneously published in The New England Journal of Medicine, highlighting its global academic and clinical value.
In terms of commercialization, Kelun-Biotech has formed a competitive initial product portfolio. Its core product, the TROP2-directed ADC sac-TMT, has been approved in China for three indications: second-line and above triple-negative breast cancer, second-line and third-line EGFR-mutated NSCLC. The HER2-directed ADC trastuzumab botidotin was approved last year for second-line and above HER2-positive breast cancer, becoming the first domestically developed HER2-directed ADC approved for this indication. Furthermore, the anti-EGFR monoclonal antibody Cetuximab N01 (达泰莱®) for RAS wild-type colorectal cancer and the anti-PD-L1 monoclonal antibody tagitanlimab (科泰莱®) for nasopharyngeal carcinoma have been launched. Another small-molecule RET inhibitor A400 is expected to be approved within the year, bringing the total number of commercialized products in China to five. Currently, three of the Company's commercialized products, covering five indications, have been included in the National Reimbursement Drug List (NRDL), further benefiting a broad population of cancer patients.
While strengthening its presence in the domestic market, Kelun-Biotech is actively expanding overseas. It has established collaborations with MSD, Ellipses, Windward Bio and Crescent Biopharma to maximize the value of its pipeline value and corporate worth. Among these, MSD is evaluating 16 global Phase III clinical studies of sac-TMT.
The breakthroughs in both clinical development and commercialization are driven by the Company's sustained investment in innovative R&D. With over a decade of accumulation in the ADC field, Kelun-Biotech's proprietary OptiDC™ platform enables differentiated design of drug candidates, which combines specific targets or targeting mechanisms with the most suitable payload-linker strategy to balance efficacy and safety. Additionally, the company is adopting a "multi-pronged" innovation strategy to continuously enhance platform capabilities. By exploring novel targets, new payloads, and diverse conjugation technologies, it is expanding the application boundaries across both oncology and non-oncology fields.
Looking ahead, Kelun-Biotech will consolidate its foundations in R&D, technologies, platforms, and operations by Executing five key development strategies. Concurrently, the Company will elevate its globalization strategy, enhancing its capabilities in product development, registration, and commercialization in ex-China market, advancing on its path to becoming a world-class biopharmaceutical company.
Please refer to the "Investor Relations-Investor Calendar" page of the company's official website for the presentation materials. You can visit the website for further details.
About Kelun-Biotech
Kelun-Biotech (6990.HK) is a holding subsidiary of Kelun Pharmaceutical (002422.SZ), which focuses on the R&D, manufacturing, commercialization and global collaboration of innovative biological drugs and small molecule drugs. Kelun-Biotech focuses on major disease areas such as solid tumors, autoimmune, and metabolic diseases, and in establishing a globalized drug development and industrialization platform to address the unmet medical needs in China and the rest of world. Kelun-Biotech is committed to becoming a leading global enterprise in the field of innovative drugs. At present, Kelun-Biotech has more than 30 ongoing key innovative drug projects, of which 4 projects have been approved for marketing, 1 project is in the NDA stage and more than 10 projects are in the clinical stage. Kelun-Biotech has established one of the world's leading proprietary ADC and novel DC platforms, OptiDC™, and has 2 ADC projects approved for marketing, and multiple ADC and novel DC assets in clinical or preclinical research stage. For more information, please visit https://en.kelun-biotech.com/.
2024-06-18
More
("Kelun-Biotech" or the "Company", SEHK: 6990.HK) announces that the Company has received a Notice of Clinical Trial Approval (IND) for SKB518, an innovative ADC drug developed by the Company, in patients with advanced solid tumors from the Center for Drug Evaluation (CDE) of the State Drug Administration (NDA). (CDE) of the National Drug Administration (NMPA) for the approval of the clinical trial application (IND) of SKB518 for injection, an innovative ADC drug developed by the Company, to conduct clinical trials in patients with advanced solid tumors.

SKB518 for injection is an innovative antibody-coupled drug with proprietary intellectual property rights developed by the Company by utilizing the "OptiDC?" platform technology in light of the biological characteristics of the target site, and it has demonstrated good efficacy and safety window in the preclinical stage.
Dr. Junyou Ge, Chief Executive Officer of Kelun-Biotech, said, "The entry of SKB518 into clinical trials enriches Kelun-Biotech's clinical product portfolio in the field of oncology ADC therapeutics, and paves a broader path for the company's flexible development of future pipelines in the oncology field. We are simultaneously accelerating the independent research and development of more than 10 preclinical-stage ADCs and novel coupling technology drug assets, and this year we plan to continue to file clinical INDs for a number of preclinical ADC pipelines."
About Kelun-Biotech
Kelun-Biotech(6990.HK)is a holding subsidiary of Kelun Pharmaceutical (002422.SZ), which focuses on the R&D, manufacturing, commercialization and global collaboration of innovative biological drugs and small molecule drugs. The company focuses on major disease areas such as solid tumors, autoimmune, inflammatory, and metabolic diseases, and in establishing a globalized drug development and industrialization platform to address the unmet medical needs in China and the rest of world. The Company is committed to becoming a leading global enterprise in the field of innovative drugs.
At present, the Company has more than 30 ongoing innovative projects in major disease areas such as solid tumors, autoimmune, inflammatory, and metabolic diseases, including over 10 projects in the clinical stage and 4 projects in the NDA stage with several global trials being conducted simultaneously in multiple countries, including China, Europe, and the United States. The company has established one of the world’s leading proprietary ADC platforms, OptiDC?, and has 5 ADC projects in the clinical stage (2 of which are in the NDA stage) and several projects in the preclinical stage. For more information, please visit https://kelun-biotech.com/.
2024-05-24
More

(May 24th, Chengdu and New Jersey) The 2024 American Society of Clinical Oncology (ASCO) Annual Meeting to be held in Chicago, Illinois, the United States of America from May 31 to June 4, 2024, Sichuan Kelun-Biotech Biopharmaceutical Co., Ltd. (the “Company”) will present two clinical stage study results at ASCO.
1.The Phase 3 OptiTROP-Breast01 study of its anti-TROP2 ADC sacituzumab tirumotecan (sac-TMT) (formerly SKB264/MK-2870) in patients with previously treated locally recurrent or metastatic triple-negative breast cancer (TNBC).
? Session: Special clinical science symposium (Abstract #104; Next-Generation Antibody–Drug Conjugates: The Revolution Continues),
? Time: June 2, 2024, 9:45 AM to 11:15 AM local time
2. The Phase 2 OptiTROP-Lung01 study of sac-TMT in combination with KL-A167 (an anti-PD-L1 mAb) as 1L treatment for patients with advanced non-small cell lung cancer (NSCLC)
? Session: oral (Abstract #8502; Lung Cancer—Non-Small Cell Metastatic Oral)
? Time: May 31, 2024, 2:45 PM to 5:45 PM local time.
Sac-TMT is jointly developed by Kelun-Biotech and MSD (the tradename of Merck & Co., Inc., Rahway, NJ, USA) at clinical stage.

The abstracts for the above studies were published on ASCO’s official website on [May 23, 2024], local time. The study results are summarized as follows:
TNBC
Patients were randomly assigned (1:1) to receive sac-TMT (n = 130) or chemotherapy (n = 133). The median age was 51 years; 87% had visceral metastases; 26% received prior PD-1/PD-L1 inhibitors; 48% received three or more prior lines of chemotherapy for advanced disease. The primary endpoint of progression free survival (PFS) was met based on interim analysis (data cut-off: Jun 21, 2023) with a 69% reduction in risk of progression or death (HR 0.31; 95% CI, 0.22 to 0.45; P <0.00001).
The median PFS, as assessed by BICR, was 5.7 months (95% CI, 4.3 to 7.2) with sac-TMT and 2.3 months (95% CI, 1.6 to 2.7) with chemotherapy; PFS rate at 6 months was 43.4% vs 11.1%. In the subset of patients with trophoblast cell-surface antigen 2 (TROP2) H-score > 200, the median PFS was 5.8 months with sac-TMT and 1.9 months with chemotherapy (HR 0.28; 95% CI, 0.17 to 0.48). At the first planned interim analysis for overall survival (OS) (data cut-off: Nov 30, 2023) with median follow-up of 10.4 months, OS was statistically significant in favor of sac-TMT (HR 0.53; 95% CI, 0.36 to 0.78; P =0.0005); the median OS was not reached (95% CI, 11.2 to NE) with sac-TMT and 9.4 months (95% CI, 8.5 to 11.7) with chemotherapy. The objective response rate (ORR) assessed by BICR was 43.8% with sac-TMT and 12.8% with chemotherapy.
Most common grade ≥ 3 treatment-related adverse events (TRAEs) (sac-TMT vs. chemotherapy) were neutrophil count decreased (32.3% vs. 47.0%), anemia (27.7% vs. 6.1%) and white blood cell count (WBC) decreased (25.4% vs. 36.4%).
A Phase 3 global study led by MSD of sac-TMT plus pembrolizumab versus treatment of physician's choice (TPC) in TNBC who received neoadjuvant therapy and did not achieve a pathological complete response (pCR) at surgery (NCT06393374) and a Phase 3 study led by the Company of sac-TMT in China for 1L treatment of unresectable locally advanced, recurrent or metastatic PD-L1 negative TNBC (NCT06279364) are ongoing.
NSCLC
Patients with treatment naive advanced NSCLC without actionable genomic alterations were enrolled to receive sac-TMT 5 mg/kg Q3W plus KL-A167 1200 mg Q3W (cohort 1A) or sac-TMT 5 mg/kg Q2W plus KL-A167 900 mg Q2W (cohort 1B) in a non-randomized manner until disease progression or unacceptable toxicity. As of January 02, 2024, 40 and 63 patients have been enrolled in cohort 1A and 1B, respectively. Median ages were 63/63 years (cohort 1A/1B); 97.5%/85.7% had Eastern Cooperative Oncology Group (ECOG) Performance status (PS) of 1; 30.0%/33.3%, 32.5%/30.2% and 37.5%/36.5% of patients had programmed death ligand 1 (PD-L1) expression < 1%, 1%-49% and ≥ 50% of tumor cells by IHC 22C3 pharmDx assay, respectively.
After median follow up of 14.0 months for cohort 1A, the ORR was 48.6% (18/37, 2 pending confirmation), disease control rate (DCR) was 94.6% and median PFS was 15.4 months (95% CI: 6.7, NE) with a 6-month PFS rate of 69.2%. After median follow-up of 6.9 months for cohort 1B, the ORR was 77.6% (45/58, 5 pending confirmation), DCR was 100% and median PFS was not reached with a 6-month PFS rate of 84.6%. Additional subgroup analyses of cohort 1B are shown in the following table:

*Including confirmed or unconfirmed response. ORR was calculated based on response evaluable population defined as patients with ≥ 1 on-study scans.
In cohorts 1A and 1B, the most common Grade ≥ 3 TRAEs were neutrophil count decreased (30.0%/30.2%), WBC decreased (5.0%/17.5%), anemia (5.0%/15.9%), rash (5.0%/6.3%) and drug eruption (7.5%/0). Treatment-related adverse events leading to discontinuation of sac-TMT occurred in 1 patient of cohort 1B due to drug hypersensitivity, and there were no treatment-related deaths.
Two Phase 3 global studies led by MSD of sac-TMT in patients with 3L+ EGFR mutant NSCLC (NCT06074588), and 2L EGFR mutant NSCLC (NCT06305754) and a Phase 3 study led by the Company of sac-TMT in China in patients with 2L EGFR mutant NSCLC (NCT05870319) are ongoing. Additionally, Three Phase 3 global studies led by MSD of sac-TMT plus pembrolizumab are ongoing: One in patients with 1L Metastatic Squamous NSCLC (NCT06422143) , a second in patients with metastatic NSCLC expressing PD-L1 ≥ 50% (NCT06170788), and the third in patients with resectable NSCLC not achieving a pathological complete response (NCT06312137) .
2024-05-20
More
On May 16, 2024, Kelun Group celebrated the Group's twenty-eighth birthday. On the occasion of the 28th birthday of Kelun Group, Kelun-Biotech (6990.HK), together with Kelun Research Institute, honored the long-serving employees and celebrated with them. The "28 years of riding the wind together, I grow with Kelun" series of staff activities came to a successful conclusion.
Activity 1: "Cultural Symbiosis" Theme Activity
The "Cultural Symbiosis" activity began with the firm and powerful "Elite Oath" of 170 employees from 34 teams. In the "Culture Lecture - Side by Side - Culture Transmission - Working Together - Dedication to Factory Day" competition, colleagues within the team support each other to work together, the teams learn from each other to catch up with each other, in a pleasant and intense atmosphere to promote the corporate culture, enhance the team cohesion, and match the style of the people of Kelun!

Highlights of the event


Activity 2: "All the Way" Long-term Service Employee Recognition Ceremony
To fight for the waves, a group of Kelun people have been working together with great expertise. In order to inspire and thank the long-serving employees of Kelun, on May 16th, the Group's birthday, Kelun-Biotech and Kelun Research Institute honored more than 40 employees who have joined Kelun Group for 15 years and above.
At the recognition ceremony, the company management presented each long-service employee with a certificate of honor and a souvenir. Employees are one of the most valuable assets of Kelun, they have traveled with Kelun all the way, not only witnessed the growth of the company, but also promoted the development of the company so far.
Kelun-Biotech also looks forward to more employees joining the ranks of long-term service in the future, witnessing the growth of Colum, meeting more challenges and opportunities together with the company, and becoming the backbone of promoting the development of the company.
"Twenty-eight years of riding the wind together, I grow up with Kelun", all the staff of Kelun-Biotech will be with good wishes for Kelun, and with the company to sail the waves together to make a new chapter!
2024-05-16
More

2024-05-04
More
(stock code: 6990.HK, hereinafter referred to as "Kelun-Biotech" or the "Company") has received approval from the U.S. Food and Drug Administration (FDA) to conduct Phase 2 clinical trials for its main product, A400 (also known as KL590586 or EP0031). (also known as KL590586 or EP0031), the Company's main product, has been approved by the U.S. Food and Drug Administration (FDA) for Phase 2 clinical trials.

A400 (EP0031) is a second-generation selective RET inhibitor (SRI) with broad activity against common RET gene fusions and mutations, designed to address clinical needs not met by first-generation SRIs. Targeting RET-driven cancers, next-generation SRIs offer potentially more diverse treatment options and may further improve patient prognosis.
In March 2021, Kelun-Biotech granted an exclusive, paid-up license to Ellipses Pharma Limited ("Ellipses"), a UK-based pharmaceutical company, to develop, manufacture and commercialize A400 (EP0031) in all countries outside of Greater China, North Korea, South Korea, Singapore, Malaysia and Thailand. (EP0031) in all countries except Greater China, Korea, South Korea, Singapore, Malaysia and Thailand.
In June 2022, A400 (EP0031) received FDA approval of an Investigational New Drug (IND) application for a Phase 1/2 trial in patients with RET-altered malignancies.
In November 2023,A400 was granted orphan drug status designation by the FDA for the treatment of RET fusion-positive solid tumors.
In March 2024, A400 was granted Fast Track designation by the FDA for the treatment of RET fusion-positive non-small cell lung cancer (NSCLC).
Currently, Kelun-Biotech is conducting a pivotal clinical study of A400 (EP0031) in China in RET-positive NSCLC.
In preclinical studies, A400 (EP0031) demonstrated favorable inhibitory activity against major RET kinases in vitro and in vivo, and A400 (EP0031) also demonstrated good blood-brain barrier penetration in animal models.
Data on A400 (EP0031) shared at the 2023 American Society of Clinical Oncology (ASCO) Annual Meeting showed that, based on the results of its ongoing Phase 1/2 trial, A400 (EP0031) demonstrated good anti-tumor efficacy in patients with advanced RET+ solid tumors, particularly in first- and second- or higher-line advanced RET+ NSCLC, with ORRs of 80.8% and 69.7%. The DCR in both cases was reported to be more than 96%.
At the upcoming 2024 ASCO Annual Meeting, the Company's partner Ellipses will report clinical data from the A400 (EP0031) Phase 1 dose-escalation and extension study conducted in patients with advanced RET variant NSCLC and other oncology treatments who have never been treated with an SRI or prior therapy on June 3, 2024 (local time).
Kelun-Biotech is committed to developing innovative medicines with features and advantages to address global unmet clinical needs, and the company will also accelerate the development of the drug in China and work with its partner Ellipses to advance the global development and commercialization of A400 (EP0031) to benefit more oncology patients around the world.
About Kelun-Biotech
Kelun-Biotech(6990.HK)is a holding subsidiary of Kelun Pharmaceutical (002422.SZ), which focuses on the R&D, manufacturing, commercialization and global collaboration of innovative biological drugs and small molecule drugs. The company focuses on major disease areas such as solid tumors, autoimmune, inflammatory, and metabolic diseases, and in establishing a globalized drug development and industrialization platform to address the unmet medical needs in China and the rest of world. The Company is committed to becoming a leading global enterprise in the field of innovative drugs.
At present, the Company has more than 30 ongoing innovative projects in major disease areas such as solid tumors, autoimmune, inflammatory, and metabolic diseases, including over 10 projects in the clinical stage and 4 projects in the NDA stage with several global trials being conducted simultaneously in multiple countries, including China, Europe, and the United States. The company has established one of the world’s leading proprietary ADC platforms, OptiDC?, and has 5 ADC projects in the clinical stage (2 of which are in the NDA stage) and several projects in the preclinical stage. For more information, please visit https://kelun-biotech.com/.
2024-04-12
More

In this beautiful season of "wet with apricot blossom rain, not cold with willow wind", the work conference of 2024 Kelun-Biotech was grandly held in Chengdu from April 10th to 12th. Mr. Liu Gexin, Chairman of the Board, Mr. Liu Sichuan, General Manager of the Group, senior management team, relevant persons in charge of each subsidiary (branch) company, each marketing area and all departments directly under the company attended the meeting, and at the same time, the whole meeting was broadcasted in the form of video, with a total number of more than 4,000 people attending the meeting. According to the agenda of the meeting, the 11th was synchronized with the meeting of the industrial sub-groups and marketing sub-groups, and the morning of the 12th was the working conference of the group. A total of 39 speakers made presentations and focused on the short-term and future trends of production and marketing.
The year of 2023 is the year when Kelun resolutely implements the "Twenty Character Guidelines" and continues to move forward victoriously, and the vision and courage of all the fighters extend the road of success of Kelun here. The successful spin-off and listing of Chuanning Biologicals and Kelun-Biotech, Kelun has formed a "character" structure operation platform, and as of March 2024, the total market capitalization has exceeded 100 billion yuan, becoming a pioneering force to lead the industry's high-quality development.
The industrial sub-group meeting focused on safety, quality, cost and other core themes. Kelun Drug Research Institute, Xindu Base, Hunan Kelun, Yueyang Branch, Hubei Kelun, Qionglai Branch focused on research and production linkage, production and marketing linkage, intelligent manufacturing, and high-quality development were comprehensively summarized.
The EHS Supervision Department, a department directly under the headquarters, focused on problems, prevented small steps, strengthened the construction of the safety system and the implementation of responsibilities, and continued to optimize the safety system and strengthen the safety culture with the help of PMS and other management tools. The Cost Management Committee put forward the company's thoughts and requirements on cost management from optimizing cost management, efficient linkage cost reduction, etc., and formulated a cost-saving incentive program, which injected a powerful agent for the production enterprises to continuously reduce costs and increase efficiency. The General Manager's Department made an in-depth summary around the dimensions of solidifying the quality cornerstone, creating an economic supply center, innovation empowerment, and improving quality and efficiency, and made important work plans and deployments in promoting industrial upgrading, and creating a competitive advantage in quality and cost.
The agenda of the meeting was also interspersed with thematic discussion sessions, for the current hotspots and production quality management practices, Internet + safety production, material key attribute assessment and control, supplier control strategy, artificial intelligence and other in-depth discussions, clarifying the management ideas, clearing the direction of the promotion and strategy.
At the end of the theme report, Mr. Liu Gexin, the chairman of the board of directors, commented on the speeches of each theme and pointed out that: soldiers are precious and quick! In the current market environment, through the adjustment of industrial structure, rapid release of production capacity, stabilize market expectations; follow the "Hein's Law", cast product quality shield.
Since its establishment in 2021, under the guidance of the company's "twenty-word policy", Kelun Marketing Center has further improved the company's operational efficiency with reform thinking and innovative approaches. At present, the marketing center is at the intersection of two three-year plans, this meeting comprehensively summarized the first three-year plan and comprehensively deployed the tasks of the second three-year plan.
Core Business Marketing Division, New Drug Marketing Division, Kelun-Biotech Marketing Center, CDMO Business Development Department, Comprehensive Products Division and Large Retail Division analyzed and summarized the sales and market situation of the company's core infusion products and generic innovative products from 2021 to 2023, and put forward the sales planning for 2024 and the long term; Health Strategy Development Research Department, County Business Department, Merchants Department and excellent areas shared the system construction, academic promotion and other multi-dimensional work experience. The Market Access Department, the Integrated Medical Market Department and the Business Department made special reports on the layout of access work, the professional capacity and system construction of academic promotion, and the management of product channels.

After listening carefully to the special report of each spokesman of the marketing sub-group, General Manager of the Marketing Center Fan Wendi made an in-depth and detailed comment, and deployed the second three-year plan of the Marketing Center - Deepening Reform, Steady Transformation, from the four dimensions of organizational optimization and talent cultivation, deepening of the business and comprehensive integration, improving the effectiveness and compliance transformation, and opportunities and problems. He also pointed out that China's pharmaceutical industry has gone through the dilemma of "no drugs available", the market-oriented operation of pulling up seedlings to help them grow, the difficult period of domestic substitution of generic drugs, and the agitation under the innovation dividend, and is now moving towards rationality and maturity. Every day, we must find the long-term trend in the changes and explore new breakthroughs in the unchanged!
Chairman Liu Gexin instructed: the top management has reached a high degree of consensus on the upgrading and transformation of marketing; the ice has been broken, the course has been indicated, and the road has been opened, and we are going to take resolute actions to quickly reach the expected reform goals.

Mr. Deng Xuheng, General Manager of Chuanning Bio, said: Chuanning adheres to the innovative development concept of building industrial alliance in known fields and forming knowledge alliance in unknown fields, and has been at the forefront of the industry in terms of industrial scale, product quality, environmental protection and governance level, and has become the head producer with international discourse power in bulk pharmaceutical raw materials. After listing, Chuanning has focused its resources on the synthetic biology track and completed the research and development of its first synthetic biology product, red myrcene glycol, to commercial production in two years, which fully demonstrated Chuanning's high-quality research and development level and strong industrialization capability. In the future, Chuanning will continue to adhere to the dual-wheel drive strategy of "bio-fermentation" and "synthetic biology", empowering the antibiotic industry and synthetic biology R&D with AI to further consolidate the head position of the traditional antibiotic industry, and at the same time accelerating the research and development and landing of new products, which will contribute to the profitability of Coren Group. At the same time, we will accelerate the research and development of new products, and contribute to the sustainable growth of the profit of Kelun.

On behalf of Kelun Research Institute and Kelun-Biotech, Mr. Ge Junyou, General Manager of Kelun-Biotech, reported the progress of Kelun's drug R&D work in 2023 and the work objectives in 2024, taking into account the current domestic and international regulations and competitive situation. He said: At present, the challenges and opportunities of new drug R&D coexist, Kelun will continue to adhere to the R&D strategy of "Generic drives innovation, innovation drives the future", under the strong and wise leadership of the Chairman, General Manager and the Group, continue to strengthen the full-cycle empowerment of innovative drug R&D by AI technology, focus on key indications and core technology platforms, fully promote international cooperation, and accelerate the development of multiple innovative drugs, as well as the development of new drugs in the future. We will also make every effort to promote international cooperation, accelerate the approval and commercialization of many innovative drugs, maximize the global value of our pipeline, and contribute China's power to the cause of human health!
The awarding ceremony of "The Fifth Group Meeting of Kelun Group" was also held during the meeting, and the top ten outstanding marketing areas in terms of net income and per capita profit for the year of 2023, and the outstanding production enterprises in the "Billion Dollar Club" were awarded respectively. The loyal and brave generals of Kelun marketing business face the challenges with the mindset of accommodating all rivers and the confidence of never giving up; the team will be cohesive as one and the spirit of struggle to achieve the mission frequently show their swords, which is so awe-inspiring and courageous! "It seems to be the most rugged, into as easy but hard", Colum this growing, growing production and technology team, is the foundation and hope for the continued success of the Colum business; it is a group of people who must be cherished and cared for, no matter how it is evaluated will not be too high. We are bold and confident in realizing this year's goal! Let us work together to open a new era of carrying the glory of Kelun, move forward!

Chairman Liu Gexin attended the whole meeting, and after listening to the speeches on industry, marketing, Chuanning Biological and R&D innovation, he put forward opinions and suggestions in light of the actual work and responded to the concerns of all parties.
Chairman Liu Gexin talked about: a bird dares to sing in the fragile branch, why it does not worry about the branch will break? Because it has wings to fly, the enterprise is the same, from the moment of the birth of Kelun, the eagle is our spiritual totem, through the clouds and fog, the eyes of heaven and earth, "Kunpeng wings, ninety thousand miles, turn the shaking goat's horns", which is the ambition of Kelun.
After twenty-eight years of hard work, Coren is at a "high time" that has never been seen before in its history. We must be virtuous and prudent, cautious and fearful; we must adhere to long-termism and value investment, and become a trustworthy company by rewarding investors and society with more excellent results.
In the face of the great changes that have not been seen in a century, embracing change from the bottom of our hearts is the only way to see the opportunities in economic adjustments and business changes. The only way to cope with change is to innovate, which is the essence of entrepreneurship. Success lies not only in talent and luck, but also in the anticipation and selection of quality "tracks", which requires entrepreneurs to have higher wisdom and commitment to explain the present and foresee the future with new knowledge systems and deeper insights. Entrepreneurs' belief in technology, their pursuit of innovation and their confidence in the future are intertwined, and these are great enablers for creating a new era of business.
Whenever we start a new impact, we will seek the regeneration power of "Phoenix Nirvana" from the original civilization of Colum's entrepreneurship, and the spirit of Colum, which is mutually promoting and stimulating, has been honed through the vicissitudes of life and has been deposited in the hearts of Colum's comrades, and will explode when there is a fire.
Today, we are standing on a new starting point, has a completely different state of mind. Warm winds of May will soon blow across the land of China, this is the season belonging to us, since we have chosen a faraway place, we will travel through thick and thin, let's sing for the month of May, and pay homage to those who struggle!
2024-04-08
More
2024 American Association for Cancer Research (AACR) Annual Meeting is being held in San Diego, California, the United States of America from April 5th to 10th, 2024, local time.

Sichuan Kelun-Biotech Biopharmaceutical Co., Ltd. (6990.HK, the “Company”) will present two study results of anti-TROP2 ADC sacituzumab tirumotecan (sac-TMT, formerly SKB264/MK-2870) during the AACR meeting scheduled below. The abstracts for the studies have been published on the official website of the AACR on April 5th, 2024, local time (See link below).
1. The updated efficacy and safety results for its anti-TROP2 ADCSKB264/sac-TMT in patients with previously treated advanced non-small cell lung cancer (NSCLC) from a phase 2 study in a poster session scheduled on April 9 2024, 1:30 PM - 5:00 PM local time (Abstract Presentation Number: CT247).
2.The preliminary efficacy and safety results for its anti-TROP2 ADC SKB264/sac-TMT in patients with previously treated advanced gastric or gastroesophageal junction (GEJ) cancer from a phase 2 study as an oral presentation, which is scheduled in a session on April 9 2024, 2:30 PM - 4:30 PM local time (Abstract Presentation Number: CT038).

The study results are summarized as follows:
NSCLC
Patients with previously treated advanced NSCLC were enrolled to receive SKB264/sac-TMT at 5 mg/kg Q2W until disease progression or unacceptable toxicity (KL264-01, NCT04152499). The data cut-off date was November 22, 2023.
Five Phase 3 global studies of SKB264/sac-TMT in patients with NSCLC are ongoing. Including two Phase 3 global studies of SKB264/sac-TMT in patients with 3L+ EGFR mutant NSCLC (NCT06074588), and 2L EGFR mutant NSCLC (NCT06305754) and a Phase 3 study ofSKB264/sac-TMT in China in patients with 2L EGFR mutant NSCLC (NCT05870319). Additionally two Phase 3 global studies of SKB264/sac-TMT plus pembrolizumab in patients with metastatic NSCLC expressing programmed death ligand 1 (PD-L1)≥ 50% (NCT06170788) and resectable NSCLC not achieving pathological complete response (NCT06312137) are ongoing.
As of the Data Cut-off date, 43 NSCLC patients had been enrolled and the median follow-up was 17.2 months. 21 patients with EGFR wild type had received a median of 3 prior regimens of therapy including anti-PD-1/L1 inhibitors. 22 patients with EGFR mutant had progressed on or after TKI therapy, 50% of whom also failed at least one line of chemotherapy. Updated efficacy results are shown in the following:
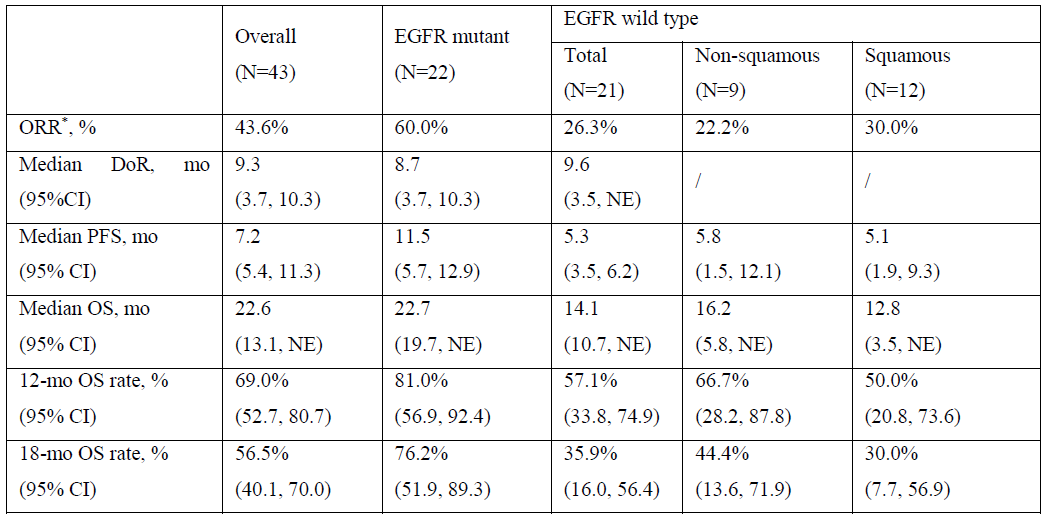
*Including confirmed or unconfirmed response. Based on response evaluable patients (≥1 on-study scans) with 4 patients (2 EGFR mutant patients with non-squamous histology and 2 EGFR wild type patients with squamous histology) excluded.
The most common Grade ≥3 treatment-related adverse events (TRAEs) were neutrophil count decreased (34.9%), anemia (30.2%), white blood cell (WBC) count decreased (25.6%), stomatitis (9.3%), and rash (7.0%). No TRAEs leading to treatment discontinuation or deaths occurred. No drug-related interstitial lung disease (ILD)/pneumonitis was reported.
Gastric/GEJ cancer
Patients with previously treated inoperable advanced gastric/GEJ adenocarcinoma were enrolled to receive SKB264/sac-TMT monotherapy at 5 mg/kg Q2W until disease progression or unacceptable toxicity in Phase 2 expansion cohort of KL264-01 study (NCT04152499). Patients with heavily pre-treated gastric/GEJ cancer were enrolled first, and then the cohort was amended to enroll patients with only one prior therapy of chemotherapy and anti-PD-1/L1 therapy. The data cut-off date was Nov 22, 2023.
As of the data cut-off date, a total of 48 patients were enrolled and followed up for at least 9 weeks. 24 patients (50.0%) had received one prior line of therapy (2L), while 24 patients (50.0%) had received ≥ 2 prior lines of therapy (3L+). 40 patients (83.3%) had received prior anti-PD-1/L1 inhibitors. Of 41 response-evaluable patients (defined as ≥ 1 on-study scans), the objective response rate (ORR) was 22.0% (9 partial responses, 2 pending confirmation) and disease control rate (DCR) was 80.5%. The ORRs in the 2L and 3L+ setting were 27.3% (including 2 pending confirmation) and 15.8%, respectively. Median duration of response (DoR) was 7.5 months. In the subset of 3L+ patients (n=24 including 54.2% of patients with ≥ 4 prior lines of therapy) with more mature follow-up (median follow up of 14.6 months), the median progression free survival (mPFS) was 3.7 months (95% CI: 2.6, 5.4) and median overall survival (mOS) was 7.6 months (95% CI: 5.3, 15.5).
The most common ≥ Grade 3 TRAEs were anemia (20.8%), neutrophil count decreased (18.8%), WBC decreased (12.5%) and neutropenia (6.3%). No TRAEs leading to treatment discontinuation or deaths occurred. No neuropathy or drug-related ILD/pneumonitis was reported.
A Phase 3 global study of SKB264/sac-TMT monotherapy versus standard of care (SOC) chemotherapy in 3L+ gastric/GEJ adenocarcinoma is being planned.
Press Contact: Xinwei Li, klbio_pr@kelun.com
2024-03-23
More

The World Meteorological Organization mentioned on the Interim Report on the State of the Global Climate 2023 that the year 2023 is the hottest year in the history of mankind since records have been kept, and the global climate is getting warmer and warmer, and the earth that we live on is facing a serious climate crisis, and energy conservation and environmental protection are imminent.

"Earth Hour was initiated by the World Wide Fund for Nature (WWF) in 2007, calling on people to voluntarily turn off their lights for one hour at the end of March each year to protect our planet.
Today, Earth Hour has become the world's largest national environmental event, focusing on the twin crises of nature loss and climate change on a global scale.
As a result of the global effort, by 2023, more than 410,000 hours have been accumulated in the "Hour Bank", an online counter for taking positive action to protect the planet.

Earth Hour 2024 will be held at 20:30pm local time on March 23, 2024, to inspire people in more than 190 countries and regions to take action to protect our common home.
Responding to the call of WWF, Kelun-Biotech actively participates in Earth Hour, and carries out all-round publicity for its employees by posting posters, broadcasting on electronic screens and sending out announcements, advocating employees to participate in the one hour lights out activity at home or in the office area from 20:30 to 21:30 on March 23rd, and encouraging employees to post Earth Hour related articles. At the same time, employees were encouraged to post Earth Hour related social media to call on their friends and relatives to participate in the activity.

Environmental protection is more than just an hour. Outside of the event period, Kelun-Biotech also actively fulfills its corporate social responsibility by practicing sustainable environmental protection and helping to move towards the environmental goal of zero load. For example, in 2023, we will save 680,000kWh of electricity by choosing energy-saving chiller units; save more than 240,000 standard cubic meters of natural gas by optimizing the air-fuel ratio of boilers; and save 35,000 tons of water by renovating some water points with recycled water.

We prefer larger or recyclable packaging, and in 2023, we will replace the outer packaging of 5L chemical reagents for R&D from cartons to plastic baskets, and replace dichloromethane and ethyl acetate from small drums to large iron drums, to effectively reduce waste emissions through control at the source.
We invite you to join us on March 23rd at 20:30 to gather the power of "One Hour", turn off unnecessary lighting, and use small actions to bring about a big change with the heart of fraternity, and build a naturally good and carbon-neutral future together.

About Kelun-Biotech
Kelun-Biotech(6990.HK)is a holding subsidiary of Kelun Pharmaceutical (002422.SZ), which focuses on the R&D, manufacturing, commercialization and global collaboration of innovative biological drugs and small molecule drugs. The company focuses on major disease areas such as solid tumors, autoimmune, inflammatory, and metabolic diseases, and in establishing a globalized drug development and industrialization platform to address the unmet medical needs in China and the rest of world. The Company is committed to becoming a leading global enterprise in the field of innovative drugs.
At present, the Company has more than 30 ongoing innovative projects in major disease areas such as solid tumors, autoimmune, inflammatory, and metabolic diseases, including over 10 projects in the clinical stage and 4 projects in the NDA stage with several global trials being conducted simultaneously in multiple countries, including China, Europe, and the United States. The company has established one of the world’s leading proprietary ADC platforms, OptiDC?, and has 5 ADC projects in the clinical stage (2 of which are in the NDA stage) and several projects in the preclinical stage. For more information, please visit https://kelun-biotech.com/.
2022年09月06日
我们决定向甘孜泸定、雅安石棉捐赠300万元现金、300万元物资,目前已成功对接甘孜州红十字会、雅安市红十字会,今天下午已经完成打款,物资根据当地所需正在紧急集结。对于灾区需要的其他支持,我们也当全力以赴。”
9月6日下午,四川科伦药业股份有限公司相关负责人告诉记者,针对四川泸定6.8级地震中受灾严重的泸定县和石棉县,他们紧急启动灾害应急处理方案,并进行现金和物资捐赠。